Differential Equations
An equation in the form of dy/dx = f(x) g(y) is called a differential equation of the first order, where f(x) and g(y) are functions of x and y respectively
E.g.
dy/dx = xy is a differential equation where x and y are functions of x and y respectively.
In order to integrate a differential equation, the variables must be separated as follows:
dy/dx = f(x) g(y)
dy/g(y) = f(x) dx
E.g.1
dy/dx = xy
∫ dy/y = ∫ x dx
ln y = x2/2 + c
y = ex2 + c
E.g.2
dy/dx = cos2y ex
dy/cos2y = ex dx
∫ dy/cos2y = ∫ ex dx
∫ dy sec2y = ∫ ex dx
tan y = ex + c
Exponential Growth / Decay
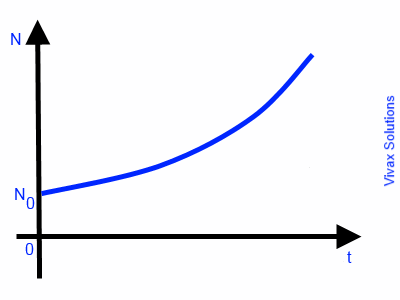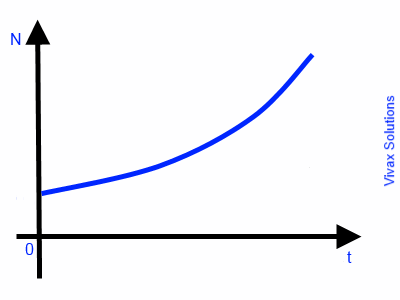
If a rate of change is proportional to its quantity, such a rate is called Exponential Growth / Decay.
E.g.1
The rate of increase in population of a colony of bacteria is proportional to the number of bacteria in the colony at a given time. Therefore, such a rate of increase is an exponential growth.
So, dN/dt ∝ N
dN/dt = k N
dN/N = k dt
∫ dN/N = ∫ k dt
ln N = kt + c
N = ekt + c
N = ekt X ec ---- 1
Let N = N0 when t = 0
N0 = ec
Sub this in 1
N = N0ekt
This is exponential growth. The following image indicates the graphical nature of the growth.
E.g.2
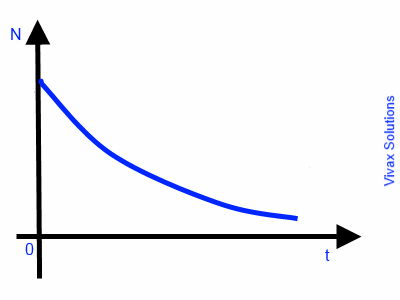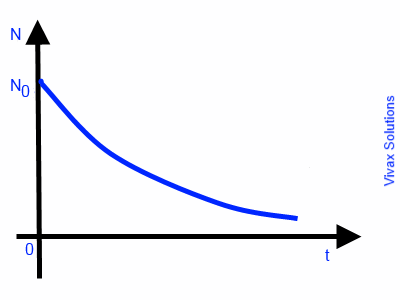
The rate of decay of radioactive nuclei in a radioactive substance is directly proportional to the number of remaining nuclei at a given time. Therefore, this is an exponential decay.
So, dN/dt ∝ -N ---- the negative sign indicates a decay or a loss
dN/dt = -k N
dN/N = -k dt
∫ dN/N = ∫ k dt
ln N = -kt + c
N = e-kt + c
N = e-kt X ec ---- 1
Let N = N0 when t = 0
N0 = ec
Sub this in 1
N = N0e-kt
This is exponential decay. The following image indicates the graphical nature of the decay against time.